Functional RNAs exhibit tolerance for non-heritable 2′–5′ versus 3′–5′ backbone heterogeneity
Aaron E. Engelhart, Matthew W. Powner & Jack W. Szostak
Nature Chemistry, 2013, 5 (5), pp 390–394. doi: 10.1038/nchem.1623
Publisher | ResearchGate | PubMed | Google Scholar
Featured in: News and Views | Nature Chemistry Editorial: “The Ascent of Molecules” | Interview with Coauthor Matt Powner | C&E News Article: “At the Origin of Life, RNA May Have Been a Mixture”
Scientific Abstract
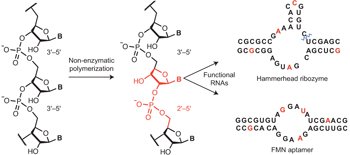
A plausible process for non-enzymatic RNA replication would greatly simplify models of the transition from prebiotic chemistry to simple biology. However, all known conditions for the chemical copying of an RNA template result in the synthesis of a complementary strand that contains a mixture of 2′-5′ and 3′-5′ linkages, rather than the selective synthesis of only 3′-5′ linkages as found in contemporary RNA. Here we show that such backbone heterogeneity is compatible with RNA folding into defined three-dimensional structures that retain molecular recognition and catalytic properties and, therefore, would not prevent the evolution of functional RNAs such as ribozymes. Moreover, the same backbone heterogeneity lowers the melting temperature of RNA duplexes that would otherwise be too stable for thermal strand separation. By allowing copied strands to dissociate, this heterogeneity may have been one of the essential features that allowed RNA to emerge as the first biopolymer.
Lay Abstract
First, a brief overview from another paper on 2′-5′ linkages in RNA, also of potential interest to readers:
Modern cells synthesize RNA with sophisticated enzymes that read a “template” strand and synthesize a second strand that is complementary – that is, where there’s an G on the template, there will be a C on the product, and where there’s an A on the template, there will be a U on the product, and so on. The enzymes that do this do so with a great deal of chemical specificity – there are actually two atoms on each nucleotide (A/G/C/U) that form part of the RNA backbone that are chemically almost the same. Despite this, the pocket inside the enzyme that catalyzes the reaction can orient the molecules in exactly the right way so that only the “right” atoms end up in the synthesized RNA strand. We can synthesize RNA in the lab without enzymes, but we aren’t nearly as good at it as the enzymes are. Depending how we set up the experiment, about 10-25% of the time, the “wrong” atom ends up in the RNA backbone, making a kink in the strand of RNA produced (the “2′-5′ linkages” – pronounced “two prime, five prime,” referring to the atoms in the ribose sugar part of RNA – mentioned in the scientific abstract – most RNA found in life has only 3′-5′ linkages). That’s about the best we can do with current technology. For a long time, these kinks have been known to have a few effects on RNA, including destabilizing the double helix structure formed between two strands of RNA, and also being less chemically stable when they’re in a double helix.
What we did in this paper was examine something that hadn’t been done before with regard to 2′-5′ linkages – look at them in functional RNAs. Since, as many people believe, RNAs might have acted as enzymes and receptors in early life, we wanted to see whether these modifications to the backbone impacted this function. It turns out, if you only have the 10-25% of unusual linkages we expect from model prebiotic reactions, RNA enzymes and receptors work just fine – a little worse than their counterparts made of regular RNA, but they are still functional. Furthermore, we showed that that small amount of 2′-5′ linkages enables us to separate two strands of RNA by heating them. Under the conditions (with dissolved salts, etc.) where we do model prebiotic RNA copying reactions, the same RNA duplexes without any 2′-5′ linkages were too stable to be separated by heat, even in water approaching boiling! It’s important to be able to separate RNA duplexes (so you can make more than one copy), so it looks like a small number of 2′-5′ linkages might have not only been tolerated in the evolution of early life, but they were something that could actually have been beneficial.